
Atoms, Molecules and Ions
The Smallest Possible Piece of Cake
Imagine you are at a birthday party, and there is one big cake to feed everyone. Everyone already there gets a piece of cake. But, someone new turns up, and also needs a piece of cake. You decide that you want to share your piece with a new friend, and you cut your piece in half. Then, another new person comes to the party, and also wants some cake, you again cut whatever you have left in half and share your piece. How long can you continue cutting your piece of cake in half if new people keep coming in late? Well, the cake is actually made up of very small building blocks, called atoms which cannot be cut up with a knife. So you can continue cutting in half until you end up with one atom of cake left!
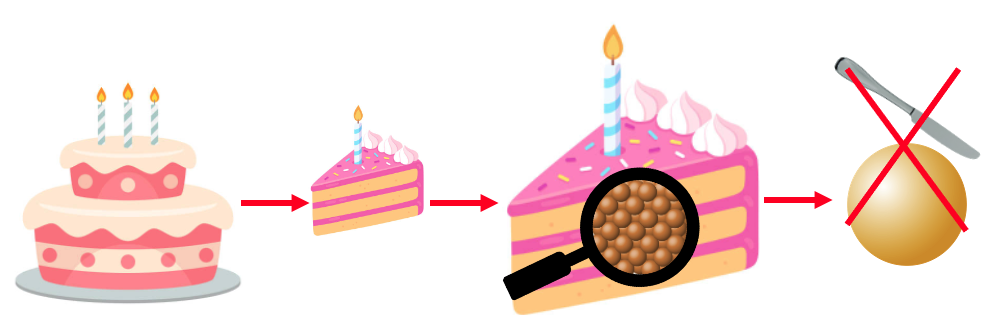
Cutting a cake until you can no longer cut. Zooming in on the last piece of cake you can see that it is made of atoms. You cannot split atoms in half and so if a new person comes in again, they won't be able to share your atom of cake.
Everything in the universe is made from atoms, not only cake. Atoms make up your body, the chair you are sitting in and the screen you are reading this from. Atoms are known as the building blocks of all matter. Matter is anything that takes up space. Atoms are very very small, they are 100 picometers across. This means that we would need 100,000,000,000,000 (one hundred thousand billion or one hundred trillion) atoms all lined up to reach one meter in length.
States of Matter
There are 4 states of matter. But we are most familiar with the first three: solids, liquids and gasses. Each of these states of matter are defined by how the atoms relate to each other in the material. In a solid, the atoms are all lined up in a rigid formation that can't easily be changed. In a liquid, the atoms are still very close together, but they can slip and slide by each other and fill the bottom of a container. In a gas, the atoms do not need to be touching each other so they fly around and fill the container they are in completely. In daily life, we come across all the three states of water: ice, water and steam. The fourth state of matter is called plasma, and we will talk about that a little later.
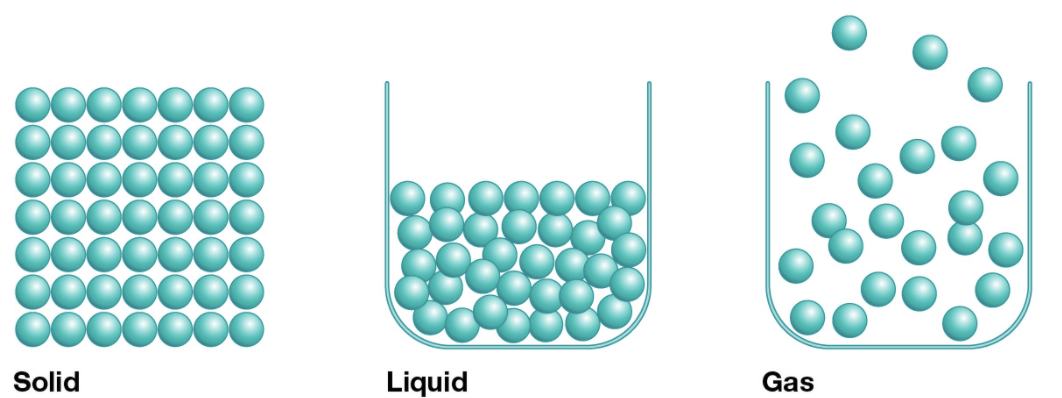
Atoms in solids liquids and gasses. Credit: Modified from Encyclopedia Britannica 2011
Electrons, Protons and Neutrons
Atoms themselves are not the smallest parts of the universe. It's just much harder to divide the parts of an atom than it is to slice a piece of cake. Inside an atom, we get electrons, protons and neutrons. Protons and neutrons stick together in a small bundle right at the center of the atom which we call the nucleus. Whereas, electrons move very quickly around this nucleus, in orbits similar to how the planets orbit around the Sun.
The nucleus makes up a very small volume inside the atom, if we scale an atom to be the size of a football stadium. The nucleus will be the size of a football on the field. Hence, an atom is actually made up of a lot of empty space in which the electrons are orbiting around
Every element (oxygen, iron, carbon...) in the universe has it's own specific number of protons and neutrons that make the atoms have the properties of the specific element. Starting with Hydrogen, which is a single proton with a single orbiting electron. Carbon, the main element in our bodies, is made up of 6 protons and 6 neutrons, with 6 electrons orbiting around. The element is defined by the number of protons, so some elements can have different numbers of neutrons and still be the same element. For example, we can also get Carbon-13 which is where there are 6 protons but 7 neutrons! We call this an isotope!
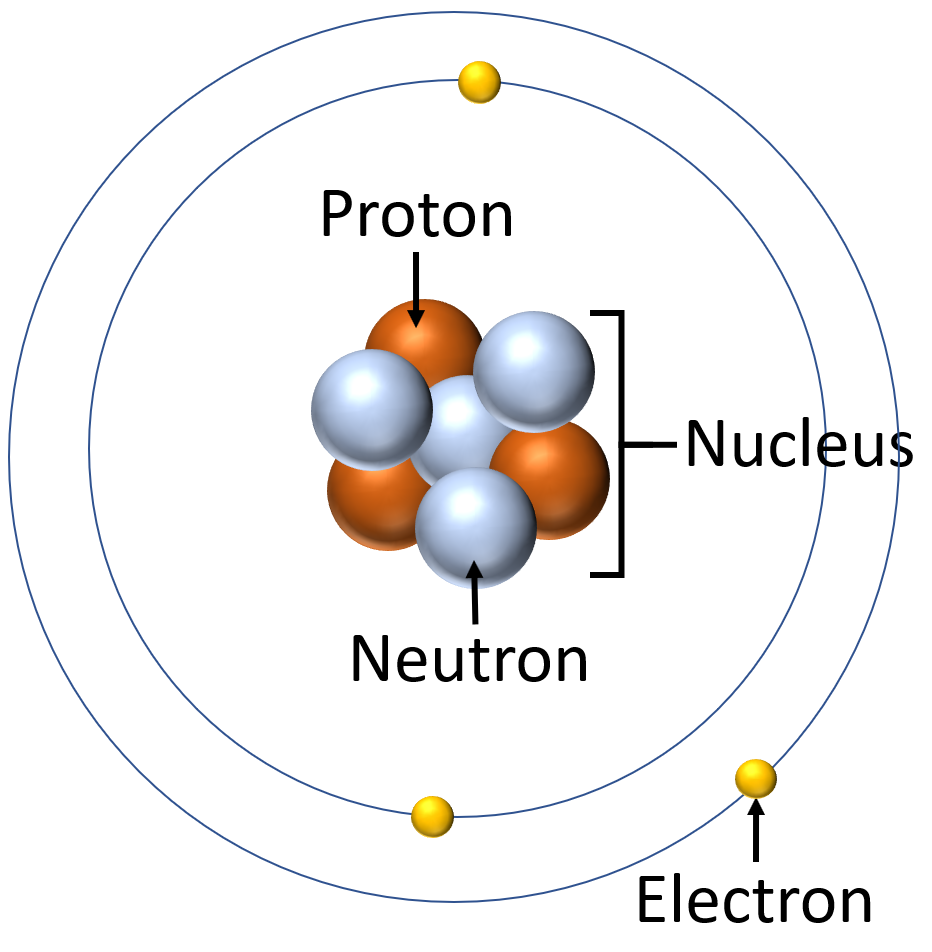
The components of an atom. Credit: keystagewiki.com
Sharing Electrons
Not everything is made up of single atom elements, some materials are made from different atoms which are stuck together to form molecules. Examples of molecules are water (hydrogen and oxygen atoms), salt (sodium and chlorine atoms) and methane (hydrogen and carbon atoms).
Some atoms like to be in molecules because atoms like to have specific numbers of electrons, and when possible, they will share electrons to make each other more comfortable or stable - meaning that they don't go around reacting with everything they touch. The number of missing electrons in each atom will dictate how many other atoms it can share electrons with, for example, a carbon atom has 4 missing electrons slots, but hydrogen only has one, hence to share and complete each other, carbon will need to find 4 hydrogen atoms to share with and become stable.
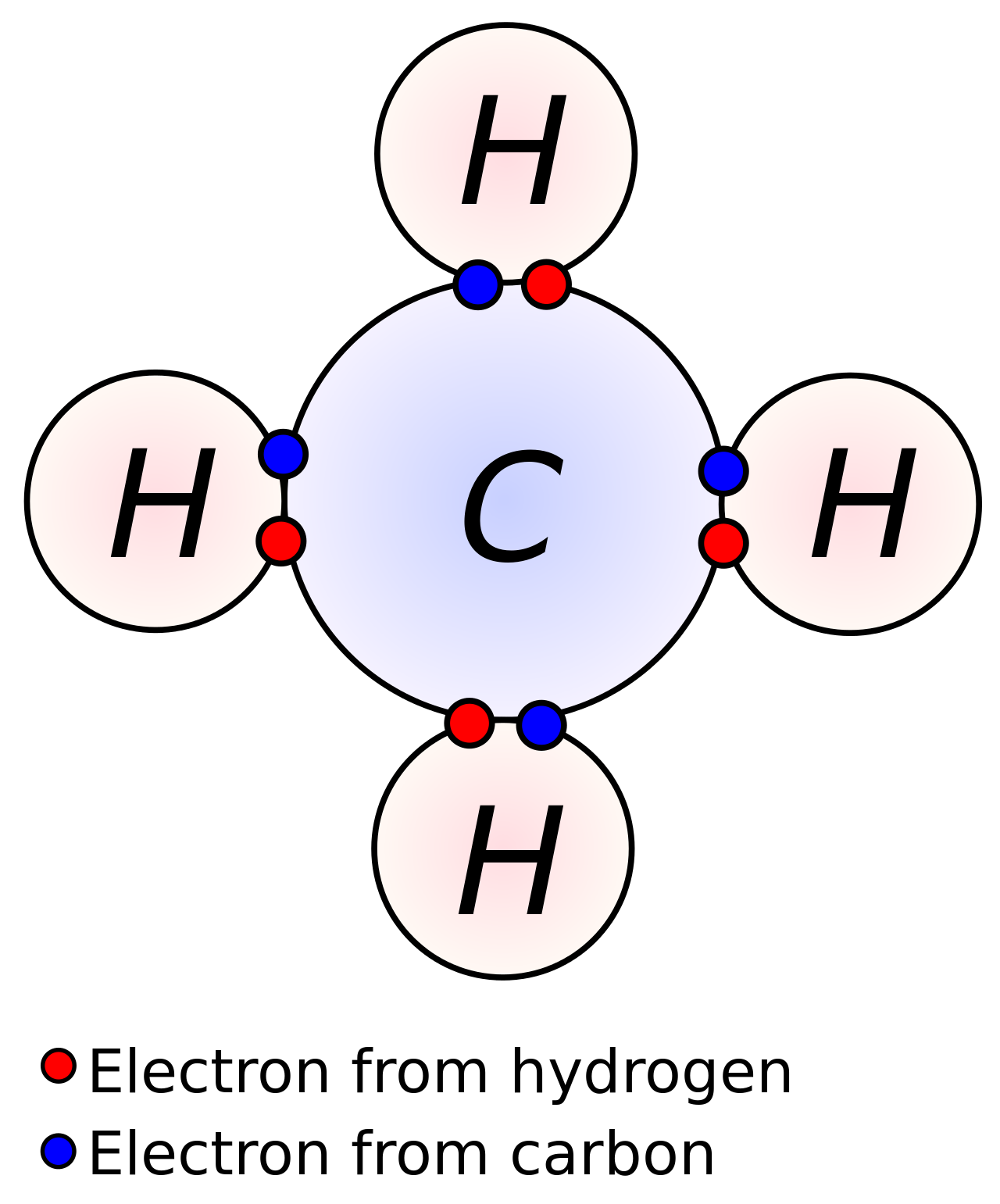
Carbon sharing 4 electrons with 4 hydrogen atoms. Credit: DynaBlast
Gaining or Losing Electrons
Protons and electrons have a property called charge. There are two types of charge that any particle can have, positive and negative. Protons have a positive charge, and electrons have a negative charge. In an atom, there are the same number of protons to electrons, and as such their charges cancel each other out, and end up with no charge on the atoms as a whole. However, in some cases atoms can fully gain or lose some electrons, rather than sharing them.
If an atom loses an electron, then there will be one more positive particle than negative, and we end up with a positive atom, or an ion. If an atom gains an electron, then it has an extra negative charge, and so the whole particle will be negatively charged. Ions are found in many things, such as batteries and the aurora borealis. In our 'states of matter' section we discussed the fourth state of matter: plasma. Plasma is formed when atoms or molecules in a gas become a gas of ions and electrons with positive and negative charges respectively!
At SuperDARN, ions are very important to our research, as they make up a large area of the atmosphere. This area of the atmosphere is called the ionosphere and we use the fact that the atoms have gained and lost electrons to track how the atoms are moving.




