
Earth's Ionosphere
What is the Ionosphere?
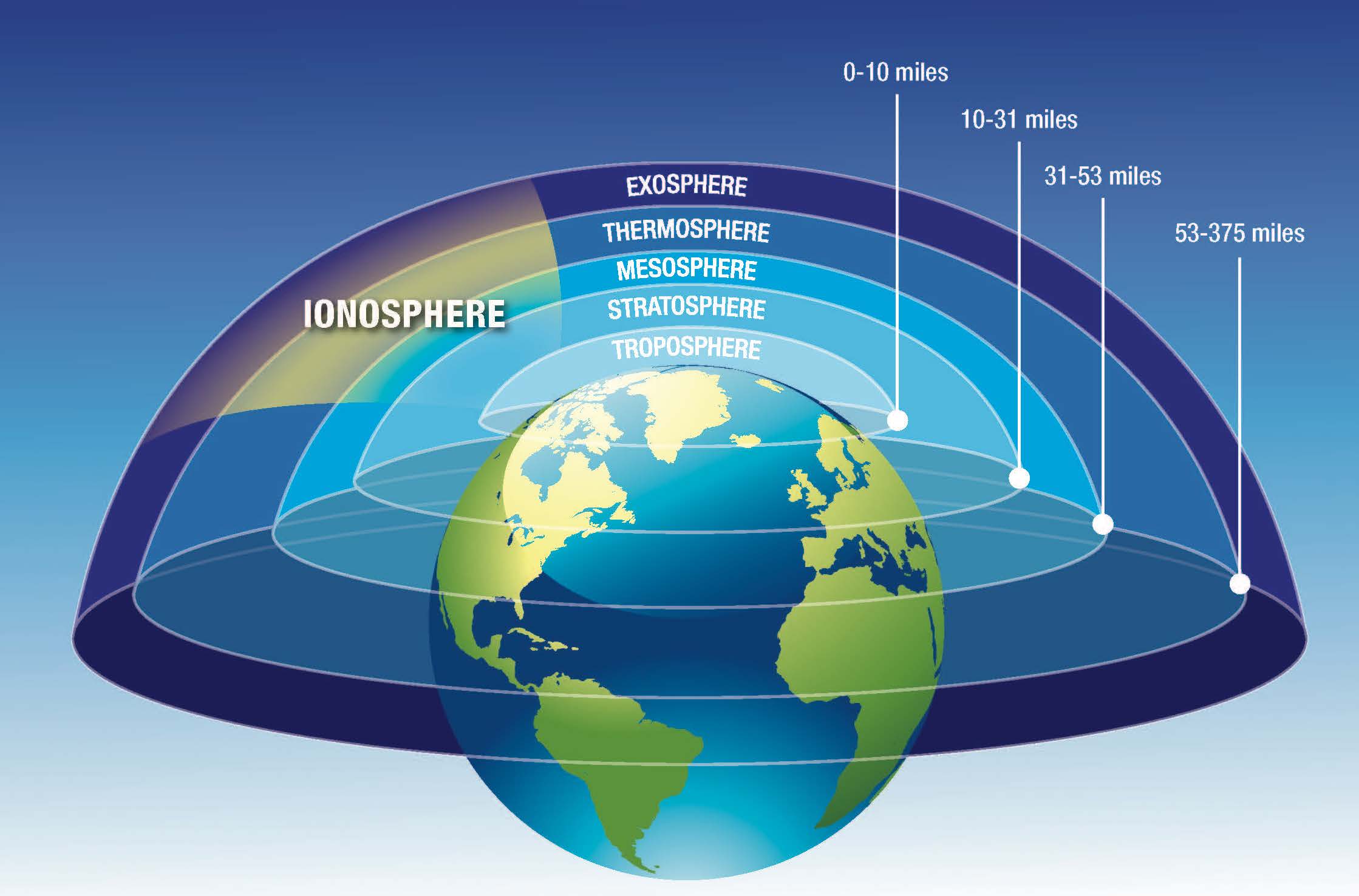
The ionosphere is an ionized part of the upper atmosphere of the Earth. We find the ionosphere from around 50 to over 1000 kilometers in height above the Earth's surface. The atmosphere that we all know and breathe, is what we call the neutral atmosphere, but when these molecules and atoms interact with various sources of ionization (the main source here being solar radiation) the electrons in these molecules are removed. As an electron is negatively charged, removing it from a neutral atom or molecule will make that atom or molecule positively charged: an ion! This leaves free moving electrons and ions, and as they are both now charged they can help electric currents flow, similar to free electrons moving charge in an electrical wire.
Scottish physicist Robert Watson-Watt first used the term 'ionosphere' in 1926, however it was not officially published until 1969.
Sunlight breaks atmospheric molecules apart, knocking off electrons and leaving behind a sea of charged electrons and ions. This population of electrically charged particles is the ionosphere, and it exists in the same space as the neutral upper atmosphere. Credit: NASA's Goddard Space Flight Center/Mary Pat Hrybyk-Keith
Ultraviolet and X-ray radiation from the Sun is classed as 'ionizing' radiation. This means that these wavelengths of light have sufficient energy to remove electrons from atoms and molecules when they interact (this is why we wear sunscreen on sunny days) and become an ion. The opposite process, recombination, is when an electron is caught by an ion, and they combine to reform the neutral atom or molecule. The balance of ionization and recombination in our ionosphere is what helps sustain the amount of free electrons present in the atmosphere.
The ionosphere was first postulated by Carl Friedrich Gauss in 1839, it wasn't until 62 years later in 1901, that the first trans-atlantic radio signal was sent from Cornwall in the UK, bounced twice off the ionosphere, and was received in Newfoundland (modern day Canada). The message sent was the morse code letter S, revolutionising wireless communications.
What is the Ionosphere Made of?
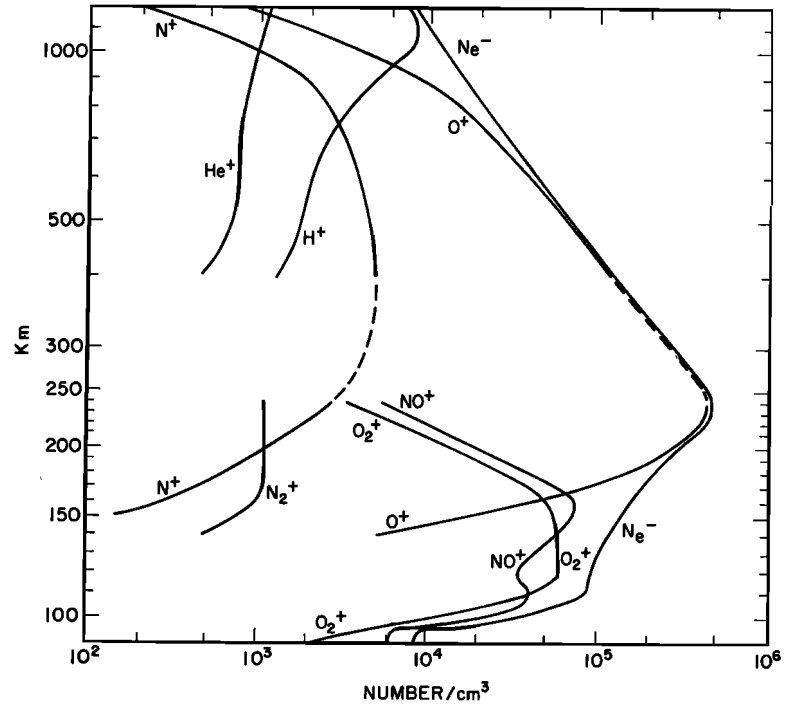
Credit: Fig 1 of Johnson, C.Y. 1966: Ionic composition of dayside ionosphere from 1963 and 1964. The x-axis depicts a log-scale of the number of ions per squared centimeter. The y-axis is a log-scale of the height in the atmosphere.
As mentioned above, the ionosphere is made up of ions and electrons which are made when the neutral atoms and molecules are ionized. So it stands to reason that the dominant species of the ionosphere would be ions of the most abundant molecule or atoms. Well, the most abundant neutral species are nitrogen and oxygen, however the most abundant ions, changing with altitude, are hydrogen and oxygen ions with some nitrogen ions.
Singly charged oxygen ions are most abundant throughout the ionosphere, with singly charged hydrogen ions only becoming dominant in the very top reaches. Heavier ions are found lower in the ionosphere as they have a greater gravitational force acting on them. Below 250 kilometers, we can find ionized oxygen and nitric oxide molecules, where below 150 kilometers these molecular ions are dominant.
The figure (left or above) shows the different ions and their abundances at different altitudes. Both axes are log axes, for example at 800 kilometers, there is roughly 10 times more oxygen (O+) than hydrogen (H+).
Ionospheric Layers
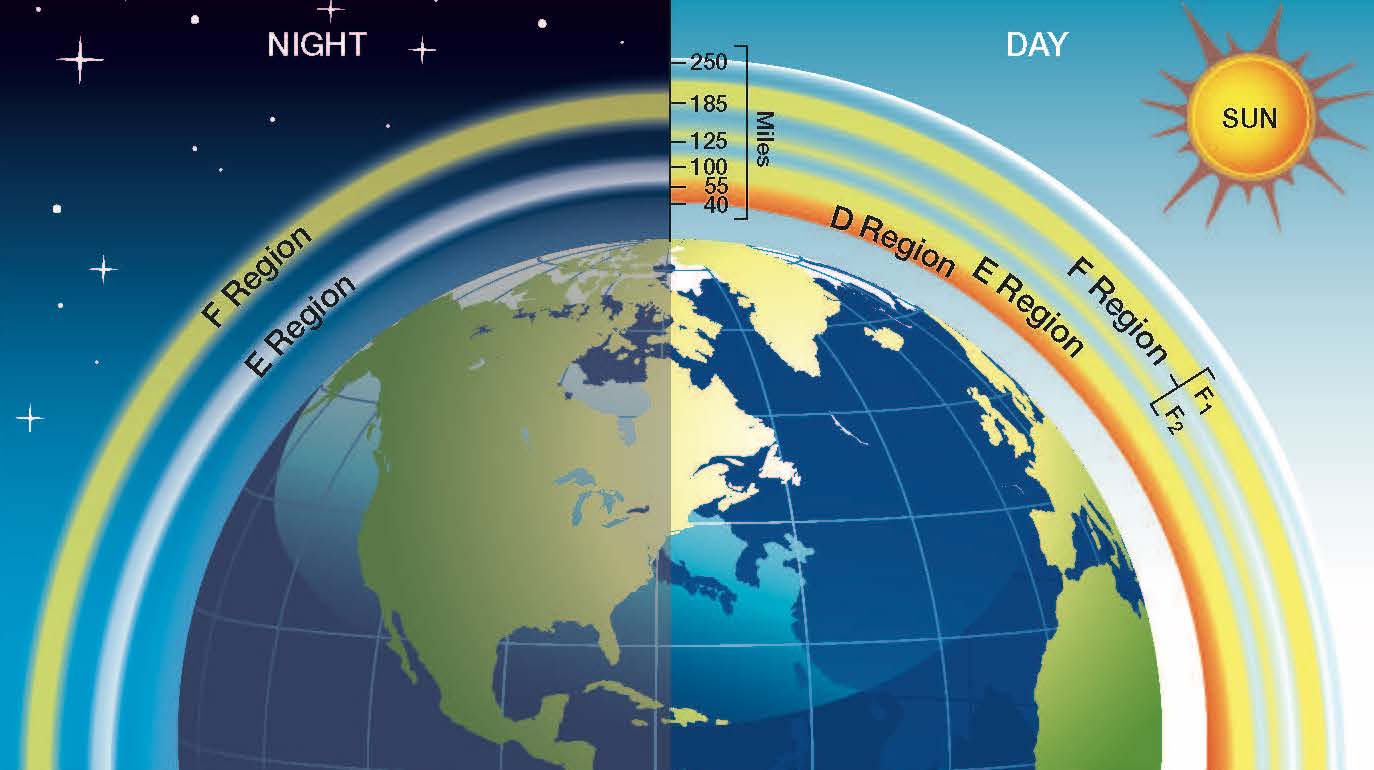
The ionosphere has a number of distinct layers (or regions), each with varying different sources and dynamics. The layers were named alphabetically, the D, E and F layers. Where the F layer can have two sub-layers, the F1 and F2 layers.
The first discovered ionospheric layer was called the D-layer, just in case any layers below were discovered. No inner layers were ever discovered, so today we still start with D!
The D-region is the lowest ionospheric layer at Earth. We can find the D-region at between 60 and 90 kilometers. The D-region is primarily caused by X-ray and ultraviolet radiation from the sun ionizing the gasses in the atmosphere. Because of this, the D-region is primarily found in the day-time ionosphere (when the atmosphere is lit up by the sun), but some residual ionisation can persist for much of the night time.
The D-region can usually absorb or attenuate low and medium frequency radio wavelengths, but allows higher frequency radio waves to pass through unhindered. At night however, because of the much lower electron density, low and medium wavelengths can pass through.
At around 100 to 125 kilometers in height above the Earth's surface, we find the E-region. This region is where our radio broadcasting waves are sometimes 'bounced' back to earth. In the Radio and Radars tutorial series, we describe what refraction and reflection mean to a radio wave. This region of the ionosphere refracts radio communications, which means that the wave's direction is changed upon entering the region, until it is changed enough that it is now moving back down towards the surface, the wave appears to be 'reflected'.
In the E-region, residual electron density can exist after dark. However, electrons and ions in the E-region recombine quickly to form a neutral gas soon after sunset, but the residual density can still attenuate some high frequency radio communications.

Credit: NASA's Goddard Space Flight Center/Mary Pat Hrybyk-Keith
The main difference between the E- and D-layers, is the surrounding atmosphere. In the D-region, the atmosphere is much denser, and as such collisions between particles is much more common. This means that in the E-region, an electron that interacts with a radio wave is much more likely to re-radiate the energy rather than lose it to a collision. Hence, the E-region refracts and the D-region absorbs radio wave communications.
Even further up in the atmosphere, at an altitude of 150-400 kilometers, is the F-region. The F-region is the layer with the highest electron density, and as such is the most important for high frequency radio signals, including us here at SuperDARN. This layer is formed mainly from atomic oxygen being ionized by extreme ultraviolet radiation from the sun.
As we climb higher in the atmosphere the neutral density becomes gradually lower than in the E-region, collisions are less frequent, and as such even less attenuation happens, meaning that this is where high frequency radio waves are refracted back to Earth. Another side effect of having few collisions, is that the electrons and ions can't bump into each other and recombine as easily. So this layer remains, even after the sun has set.
Previously mentioned, are the two stratified layers of the F-region. The two layer structure is most pronounced in the day time in Summer, when at night the two layers combine to form a single F-region. In the day time, the F1 layer is below the F2 layer, and appears as a small bump or inflection point around 300 km in a plot of electron density with altitude.The day-time/Summer splitting of the F-layer is believed to be due to the expansion of the atmosphere to higher altitudes as it heats up. As the atmosphere is hotter, heavier atoms and molecules which are easier to ionize are found higher, allowing for the F2 region to rise higher and appear to split from the F1 layer.
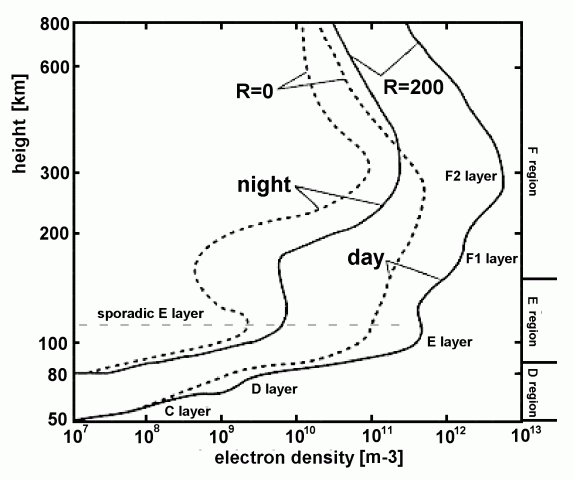
An electron density distribution for night and day. Credit: roma2.rm.ingv.it/en/research_areas/4/ionosphere
Above the F-region is what we call the Topside ionosphere. Any radio wave that makes it through to here, has already traversed the most dense part of the ionosphere, and as such the signal here is most likely lost into space. As this region is anything above the F-region, it can extend up to 800 to 1000 kilometers in altitude, until the major components of the ionosphere are the lighter Hydrogen ions. Above this is the plasmasphere, which we will discuss in the space weather tutorial series.
Changes in the Ionosphere: Perturbations and Variations
As mentioned previously, our ionosphere is sustained by a balance of ionization and recombination. This balance can be effected by a great many things. We have already discussed the changes in the ionosphere due to the daily cycle of day and night. Where the solar radiation kick-starts ionization, but when the sun sets the rate of recombination surpasses the rate of ionization and the electron density decreases, in some cases until the whole layer does not exist.
During the solar cycle (see The Sun tutorial series), the flux of radiation from the sun varies, and as such we see a knock on effect of this in the ionization rate at Earth.
Credit: NASA's Goddard Space Flight Center/Mary Pat Hrybyk-Keith
We also see a similar variation with seasons, where in the summer months the atmosphere is exposed to the solar radiation for far longer and with more strength and as such we see a much higher ionization rate in all regions. You might also think that this could persist longer into the night time, however, changes in the neutral atmosphere between winter and summer in the F-region mean that the recombination rate is far higher in the summer, meaning that we don't actually see higher electron densities in the F-region in the summer, but we see them in the winter! The E- and D-regions do follow the general increase of elections in summer though.
Your latitudinal position on Earth can also effect the ionosphere above you. For example, at the poles there is much less solar radiation, lowering the ionization rate, and vice-versa at the equator. There are many special anomalies and perturbations that affect the ionization and recombination rates. The major perturbations are due to geomagnetic storms, which we will cover in the space weather tutorial series!
Lightning can cause perturbations in the ionosphere in the D-region through LEPs or lightning-induced electron precipitation events. This is where lightning creates low frequency waves which propagate out to the radiation belt in Earth magnetosphere. The waves dislodge electrons which then precipitate or move into the D-region ionosphere, increasing electron density!
Ionospheres Beyond Earth
All of the planets in our solar system except Mercury have an ionosphere, or at least a layer of charged particles to some degree. Even though Mercury is so close to the Sun, a source of ionizing radiation, the planet's atmosphere is so thin that it cannot support an ionosphere. On Venus however, the atmosphere is very thick and hot. Even though Venus doesn't have a magnetic field of it's own, it's thick atmosphere and proximity to the Sun produces a stable and very thick ionosphere of primarily oxygen ions made from the carbon dioxide in the atmosphere. This ionosphere makes an induced magnetic field which protects the atmosphere from erosion by the solar wind.
Like Mercury, Mars has a hard time maintaining and keeping a thick atmosphere, but it does manage to produce an ionosphere, sometimes even stratified layers similar to Earth. But unlike Earth, Mars' ionosphere is sometimes influenced by dust swept up in storms on the surface, along with traces of metals. This shows that the Martian ionosphere is maintained by meteor strikes.

Jupiter’s southern aurora captured in the light of H3+ by the JUNO infrared imager. Credit: NASA/JPL-Caltech/SwRI/ASI/INAF/JIRAM
Jupiter and Saturn both have very strong magnetic fields that protect their atmospheres from erosion. Hence, they produced stable ionospheres. Both planets are made up of hydrogen and helium, and hence their main ionospheric constituents are hydrogen ions, with single ionized hydrogen and a triple-hydrogen molecular ion being dominant. The further out into the solar system we go, the less data we have and the less we know about the planets. Neptune and Uranus, both ice giants, have only even been visited once each and very little is known about their ionospheres, only that they exist.
Not just planets maintain ionospheres, many planetary moons also have atmospheres and ionospheres, like the Saturnian moon Titan. Titan's ionosphere is considered even more complex and extended than Earths due to its complex relationship with the Sun and Saturn.
References and Reading
- Lightning-induced electron precipitation, Voss et al. 1984
- MARS IONOSPHERE: A REVIEW OF EXPERIMENTAL RESULTS AND MODELING STUDIES by S.A. HAIDER ET AL, 2011
- Canadian Alouette Satellites
- britannica.com/science/ionosphere
- swpc.noaa.gov/phenomena/ionosphere
- solarsystem.nasa.gov/news/1127/10-things-to-know-about-the-ionosphere/
- royalsociety.org/science-events-and-lectures/2019/01/advances-in-hydrogen/




